Description
Changes in bowel flora may be more marked than with cefotaxime because of the greater biliary excretion of ceftriaxone; diarrhoea may occur more often, especially in children. Biliary sludge or pseudolithiasis due to a precipitate of calcium ceftriaxone has been seen occasionally in patients receiving ceftriaxone. Similarly, deposition of the calcium salt has occurred rarely in the urine. Ceftriaxone is highly protein bound and is able to displace bilirubin from albumin binding sites, causing hyperbilirubinaemia; its use should be avoided in jaundiced neonates.
Neutropenia has been reported with most cephalosporins; a complex mechanism has been attributed to that associated with ceftriaxone. There have been rare reports of fatal haemolysis associated with ceftriaxone.
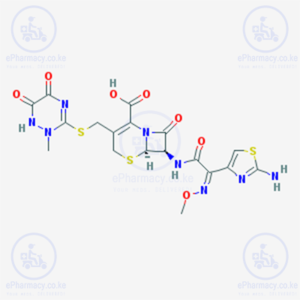
Although ceftriaxone has an N-methylthiotriazine ring rather than the N-methylthiotetrazole side-chain seen in cephalosporins such as cefamandole, it might still have the potential to cause hypoprothrombinaemia.

Reviews
There are no reviews yet.